This article was analyzed by Serge, MSc. Leveraging a background in Botany, Plant Physiology, and Biogeochemistry, I provide evidence-based insights into plant health, soil science, and sustainable cultivation. My focus is on delivering scientifically accurate data to help you grow with confidence.
People often ask if hydroponic food is actually real. There’s a common belief that soil is the only way to get ‘natural’ nutrition. But plant roots don’t have eyes, they don’t know whether they’re in a field in Iowa or a plastic bucket in a garage. What matters is bioavailability: can the plant actually absorb the minerals dissolved in the water?
The Soil Constraint and the CEC Problem
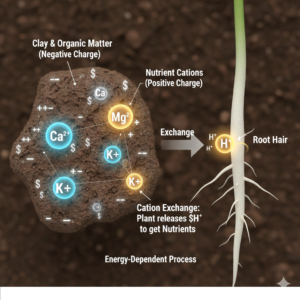
If you look at soil under a microscope, it’s a mess. Nutrients aren’t just sitting there waiting to be eaten. Most of them are locked inside organic matter or attached to tiny particles of clay. In soil science, this is described by the Cation Exchange Capacity (CEC).
Clay and organic matter are usually negatively charged, which means they attract positively charged ions like calcium (Ca²⁺), magnesium (Mg²⁺), and potassium (K⁺).
For a plant to access these nutrients, it has to literally exchange a hydrogen ion for a nutrient ion at the root surface. That process costs energy.
This is a well-evolved natural system, but it isn’t very efficient. It depends heavily on environmental conditions. If the soil gets too cold, biological activity slows down. If it gets too dry, ions can’t move. You can have extremely rich soil and still see nutrient deficiency, simply because the minerals aren’t in a form the plant can actually absorb.
Hydroponics avoids this entire limitation. The nutrients are already dissolved in the water as freely available ions. The plant doesn’t need to spend metabolic energy searching for nutrients or feeding soil microbes to release minerals. That conserved energy can be redirected toward growth, contributing to higher biomass and yield.
Stoichiometry: Why Soil Is Unpredictable
In my plant science studies, I’ve learned that stoichiometry is all about the precise balance of elements a plant needs to grow and function properly. Just like following a recipe, plants require specific ratios of nutrients, carbon (C), nitrogen (N), phosphorus (P), and trace elements to carry out essential metabolic processes.
Through my coursework and lab experience, I’ve seen that natural soils are highly variable. One part of a field might have enough iron, while another part is almost deficient, depending on how the rocks broke down over thousands of years. Even if a plant looks healthy aboveground, missing just a trace element like molybdenum can prevent it from converting nitrates into proteins, leaving it “incomplete” internally.
Hydroponic systems really highlight this contrast. By providing a controlled nutrient solution, we can ensure every essential mineral, zinc, copper, boron, and others, is available in exactly the right amounts. This helps explain why hydroponically grown vegetables often have higher vitamin content and biomass than soil-grown ones.
Recent research supports what I’ve been learning about soil nutrient balance. For example, Chen et al. (2024) studied soils under grassland, shrubland, and forestland in China’s Loess Plateau across different reforestation ages. They found that soils with higher C:N, C:P, and N:P ratios had more ammonium and nitrate nitrogen, while phosphorus availability decreased as N:P increased. Plant diversity in the upper soil layers also enhanced these ratios, showing how vegetation influences nutrient availability.
I’ve realized that soil nutrient availability is not only variable across space and depth but is also closely linked to plant-soil interactions. Understanding stoichiometric ratios helps explain why some plants thrive while others struggle, and why controlled systems like hydroponics can optimize plant growth.

The Myth of the “Watered-Down” Tomato
Many people believe hydroponic tomatoes taste bland or watery. In reality, this reputation mostly comes from large-scale commercial production, where yield and fruit weight are prioritized over flavor.
In those systems, high water availability and aggressive nitrogen fertilization can dilute sugars and flavor compounds. When nutrient chemistry is carefully managed, hydroponic produce can match or even exceed the flavor and nutritional quality of soil-grown tomatoes (Verdoliva et al., 2021).
One of the primary mechanisms involved in enhancing flavor is osmotic stress, where controlled reductions in water availability prompt the plant to concentrate sugars, oils, and secondary metabolites such as lycopene, which contribute directly to taste and nutritional value.
In field-grown systems, growers are largely dependent on weather. Heavy rain close to harvest can increase water uptake by the fruit, lowering sugar concentration and flavor intensity. In hydroponic systems, conditions are controlled. Near the end of fruit development, growers can slightly increase the electrical conductivity (EC) of the nutrient solution, reducing water availability to the plant.
This mild, controlled stress shifts plant metabolism. The plant allocates more resources toward producing sugars, oils, and secondary metabolites such as lycopene, compounds that help protect cells under stress and also contribute directly to flavor and nutritional quality. The result is a tomato with more concentrated taste and aroma!
Rather than diluting flavor, controlled hydroponic systems allow growers to manage water and nutrients precisely, concentrating what makes the fruit appealing by applying plant physiology and chemistry with intention.
The Environmental Reality Check
People see mineral salts in a bottle and think “chemicals,” which they equate to “pollution.” But let’s look at the actual biogeochemical impact of these two systems.
Water Usage: Hydroponic systems are far more water-efficient than traditional soil-based farming. Research indicates that hydroponics can reduce water usage by up to 90% compared to conventional agriculture (Rajaseger et al., 2023). In soil-grown systems, much of the water applied is lost to percolation, taking valuable nutrients with it.
In contrast, hydroponic setups recirculate nutrient solutions, delivering water and nutrients directly to plant roots and minimizing wastage. This precise management not only conserves water but also ensures optimal nutrient availability, contributing to higher yields and improved crop quality throughout the year.
Nutrient Runoff: This is a huge problem in traditional farming. Extra Nitrogen and Phosphorus wash out of the fields and into rivers, creating “dead zones” in the ocean where nothing can live. In a hydroponic system, the food stays in the tank. It’s a closed loop.
Carbon and Biodiversity: This is where soil is the hero. Healthy dirt stores carbon from the air. A hydroponic system is usually made of plastic and sits on a concrete floor; it doesn’t help the climate. We need soil to save the planet, but we need hydro to feed the people.
Bio-Hydroponics: Bringing the “Soul” to the Water
For a long time, hydroponic systems were kept very clean, using bleach or hydrogen peroxide to kill all bacteria. The goal was a “sterile” environment.
Now, scientists have found that some bacteria are actually good for plants. In bio-hydroponics, helpful microbes like Bacillus subtilis are added back into the water. These bacteria help plants take up nutrients faster and protect roots from diseases. This way, plants can grow quickly in water while keeping some of the natural taste and richness that comes from soil.
FAQs
Is hydroponic food actually healthy for humans?
Yes. In fact, it’s often cleaner. Because we control the environment, we don’t need to use heavy pesticides or herbicides that are common in soil farming. Studies consistently show the vitamin levels are either the same or higher because the plant never had a “deficient” day.
Are hydroponics better than soil?
They are different tools. If you are in a city or a desert with no water, hydro is the only way to grow. If you have a big field with healthy dirt, use the dirt. We should stop fighting about which is “better” and use both to make sure no one goes hungry.
Does soil-grown food taste better?
It can, because soil has a very complex mix of thousands of trace minerals that aren’t usually in a bottle of “A+B” nutrients. But a good hydro grower can match that flavor by adding organic acids (like Fulvic acid) and good bacteria to the water.
What about the ‘chemicals’ in the nutrients?
A Potassium ion (K+) is exactly the same whether it comes from a compost pile or a bag of mineral salt. The plant’s root has no way to tell the difference once it’s dissolved in water. The only thing that matters is that the ion is clean and available.
Can I grow anything in hydroponics?
Technically, yes. But some things are harder. Root crops like carrots or potatoes are tricky because they need something to push against to grow their shape. For greens, herbs, tomatoes, and peppers, hydro is almost always faster and heavier.
Conclusion
From my studies, I’ve realized that we don’t need to choose between soil and hydroponics. Each system has its strengths, and both can grow healthy, nutrient-rich food efficiently.
Hydroponics allows precise control of nutrients, water, and growth conditions, while soil supports natural plant–microbe interactions that are vital for long-term ecosystem health.
Understanding the chemistry of how roots absorb nutrients has shown me that we can work with natural processes while also using technology to optimize growth.
By combining knowledge from both approaches, we can produce food that uses water wisely, maintains high nutritional value, and supports sustainable production.




